Taro RX product details
Product Information
-
DIN:02380072
-
Active Ingredients :venlafaxine hydrochloride
-
Innovator Name:Effexor® XR
-
Category:Antidepressant/Anxiolytic
-
Dosage form:Capsule
-
Strength:37.5mg
- Package size:
100 Bottle 500 Bottle - UPC Code:
876515004242 876515004259 -
Non-medicinal ingredients :Black iron oxide, D&C yellow 10 (in 37.5 mg), Ethylcellulose, FD&C blue 1 (in 37.5 mg), FD&C red 40 (in 37.5 mg), Gelatin, Hypromellose, Iron oxide red, Iron oxide yellow (in 150 mg), Microcrystalline cellulose, Potassium hydroxide, Povidone, Propylene glycol, purified talc, Shellac, Sodium lauryl sulfate, Triacetin, Titanium dioxide
All brand names and trademarks referenced remain the property of their respective owners.
To view the available Product Monographs for Taro Pharmaceuticals Inc. and TaroPharma products please click here to access the Health Canada Drug Product Database and search by DIN or other listed criteria. You may also contact us to request the information.
For Provincial Formulary Status click here
Product Information
-
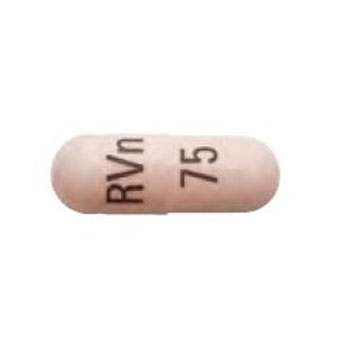
DIN:02380080
-
Active Ingredients :venlafaxine hydrochloride
-
Innovator Name:Effexor® XR
-
Category:Antidepressant/Anxiolytic
-
Dosage form:Capsule
-
Strength:75mg
- Package size:
100 Bottle 500 Bottle - UPC Code:
876515004266 876515004273 -
Non-medicinal ingredients :Black iron oxide, D&C yellow 10 (in 37.5 mg), Ethylcellulose, FD&C blue 1 (in 37.5 mg), FD&C red 40 (in 37.5 mg), Gelatin, Hypromellose, Iron oxide red, Iron oxide yellow (in 150 mg), Microcrystalline cellulose, Potassium hydroxide, Povidone, Propylene glycol, purified talc, Shellac, Sodium lauryl sulfate, Triacetin, Titanium dioxide
All brand names and trademarks referenced remain the property of their respective owners.
To view the available Product Monographs for Taro Pharmaceuticals Inc. and TaroPharma products please click here to access the Health Canada Drug Product Database and search by DIN or other listed criteria. You may also contact us to request the information.
For Provincial Formulary Status click here
Product Information
-
DIN:02380099
-
Active Ingredients :venlafaxine hydrochloride
-
Innovator Name:Effexor® XR
-
Category:Antidepressant/Anxiolytic
-
Dosage form:Capsule
-
Strength:150mg
- Package size:
100 Bottle 500 Bottle - UPC Code:
876515004280 876515004297 -
Non-medicinal ingredients :Black iron oxide, D&C yellow 10 (in 37.5 mg), Ethylcellulose, FD&C blue 1 (in 37.5 mg), FD&C red 40 (in 37.5 mg), Gelatin, Hypromellose, Iron oxide red, Iron oxide yellow (in 150 mg), Microcrystalline cellulose, Potassium hydroxide, Povidone, Propylene glycol, purified talc, Shellac, Sodium lauryl sulfate, Triacetin, Titanium dioxide
All brand names and trademarks referenced remain the property of their respective owners.
To view the available Product Monographs for Taro Pharmaceuticals Inc. and TaroPharma products please click here to access the Health Canada Drug Product Database and search by DIN or other listed criteria. You may also contact us to request the information.
For Provincial Formulary Status click here



 Taro Corporate
Taro Corporate Taro U.S.A
Taro U.S.A Taro Canada
Taro Canada Taro Israel
Taro Israel Sun Pharma
Sun Pharma Chattem Chemicals Inc.
Chattem Chemicals Inc.